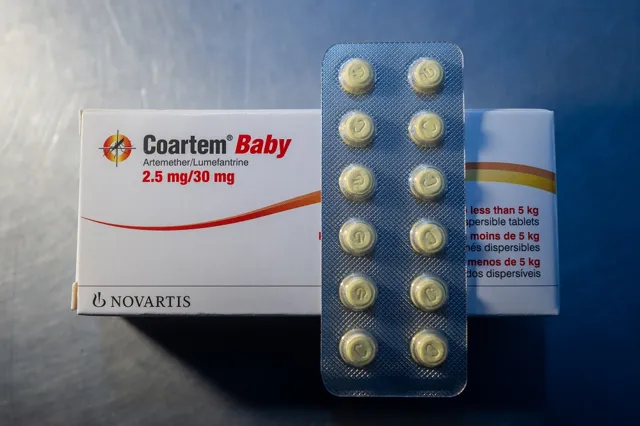
WHO Approves First Malaria Drug for Infants Under 5 kg on World Malaria Day 2026
| Aspect | Key Details |
|---|---|
| Event | WHO approves first-ever malaria drug specifically for infants under 5 kg on World Malaria Day 2026. |
| Drug Composition | Artemether-lumefantrine combination, specially formulated for newborns and young infants. |
| Significance | - Ensures safer, accurate dosing - Expedited approval in endemic countries without full local trials. |
| Regional Burden | Sub-Saharan Africa: 90% of global malaria cases/deaths; 30 million babies born yearly in endemic zones. |
| Recent Innovations | - Malaria vaccines (since 2021) - Improved diagnostics - Better mosquito nets. |
| Diagnostic Gaps | Horn of Africa: Up to 80% false-negative rates in some areas. |